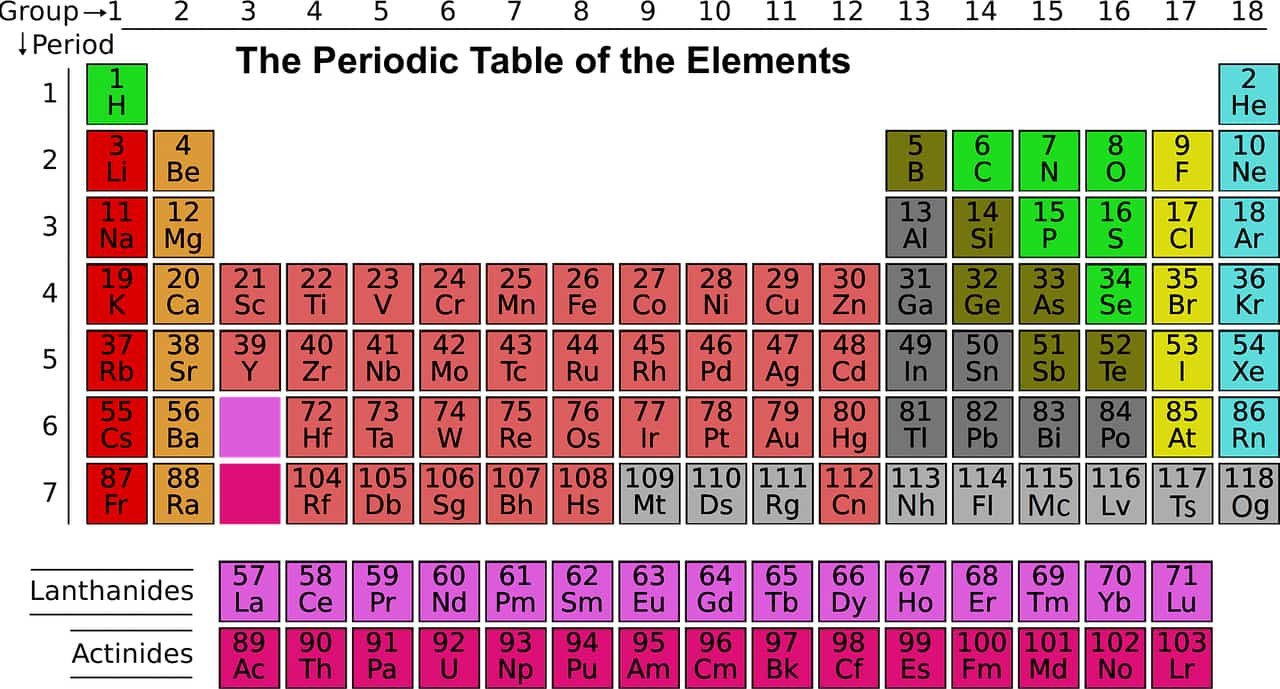
Group Periodic Table from 1 to 18
A group or family is a vertical column in the periodic table. Group periodic table usually have more significant periodic trends than periods and blocks. Modern quantum mechanical theories of atomic structure explain group trends by proposing that elements within the same group generally have the same electron configurations in their valence shell. Consequently, elements in the same group tend to have a shared chemistry and exhibit a clear trend in properties with increasing atomic number. In some parts of the periodic table, such as the d-block and the f-block, horizontal similarities can be as important as, or more pronounced than, vertical similarities. Read also: Periodic table block S P D F | The Periodic Table of the Elements
A group of the periodic table consists of the chemical elements of a single column of the periodic table. The standard array consists of 18 groups, while the columns containing the elements of block f are not numbered. The valence electrons of elements of the same group share the same electronic configuration, which gives them similar chemical and physical properties.
Under an international naming convention, the groups are numbered numerically from 1 to 18 from the leftmost column (the alkali metals) to the rightmost column (the noble gases). Previously, they were known by roman numerals. In America, the roman numerals were followed by either an “A” if the group was in the s- or p-block, or a “B” if the group was in the d-block. The roman numerals used correspond to the last digit of today’s naming convention (e.g. the group 4 elements were group IVB, and the group 14 elements were group IVA). In Europe, the lettering was similar, except that “A” was used if the group was before group 10, and “B” was used for groups including and after group 10. In addition, groups 8, 9 and 10 used to be treated as one triple-sized group, known collectively in both notations as group VIII. In 1988, the new IUPAC naming system was put into use, and the old group names were deprecated.
Elements in the same group tend to show patterns in atomic radius, ionization energy, and electronegativity. From top to bottom in a group, the atomic radii of the elements increase. Since there are more filled energy levels, valence electrons are found farther from the nucleus. From the top, each successive element has a lower ionization energy because it is easier to remove an electron since the atoms are less tightly bound. Similarly, a group has a top-to-bottom decrease in electronegativity due to an increasing distance between valence electrons and the nucleus. There are exceptions to these trends: for example, in group 11, electronegativity increases farther down the group.
Group 1 – lithium family
Group 2 – beryllium family
Group 3 – scandium family
Group 4 – titanium family
Group 5 – vanadium family
Group 6 – chromium family
Group 7 – manganese family
Group 8 – iron family
Group 9 – cobalt family
Group 10 – nickel family
Group 11 – copper family, coinage metals
Group 12 – zinc family
Group 13 – boron family
Group 14 – carbon family
Group 15 – nitrogen family
Group 16 – oxygen family
Group 17 – fluorine family (halogens)
Group 18 – helium family or neon family (noble gases)
| IUPAC group | 1a | 2 | n/a | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mendeleev (I–VIII) | I | II | III | IV | V | VI | VII | VIII | I | II | III | IV | V | VI | VII | b | |||
| CAS (US, A-B-A) | IA | IIA | IIIB | IVB | VB | VIB | VIIB | VIIIB | IB | IIB | IIIA | IVA | VA | VIA | VIIA | VIIIA | |||
| old IUPAC (Europe, A-B) | IA | IIA | IIIA | IVA | VA | VIA | VIIA | VIII | IB | IIB | IIIB | IVB | VB | VIB | VIIB | 0 | |||
| Trivial name | H and Alkali metalsr | Alkaline earth metalsr | Coinage metals | Triels | Tetrels | Pnictogensr | Chalcogensr | Halogensr | Noble gasesr | ||||||||||
| Name by elementr | Lithium group | Beryllium group | Scandium group | Titanium group | Vanadium group | Chromium group | Manganese group | Iron group | Cobalt group | Nickel group | Copper group | Zinc group | Boron group | Carbon group | Nitrogen group | Oxygen group | Fluorine group | Helium or Neon group | |
| Period 1 | H | He | |||||||||||||||||
| Period 2 | Li | Be | B | C | N | O | F | Ne | |||||||||||
| Period 3 | Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||
| Period 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |
| Period 5 | Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |
| Period 6 | Cs | Ba | La–Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Period 7 | Fr | Ra | Ac–No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
n/a Do not have a group number
b Group 18, the noble gases, were not discovered at the time of Mendeleev’s original table. Later (1902), Mendeleev accepted the evidence for their existence, and they could be placed in a new “group 0”, consistently and without breaking the periodic table principle.
r Group name as recommended by IUPAC.
Periodic Table of Elements | Complete List of Chemical Elements by Group, Name, Symbol, Color and Type
Source: Royal Society of Chemistry


